Heat and Thermodynamics
Conduction
-Conduction- The transfer of thermal energy with no overall transfer of matter.
-Conduction happens when two things are together and they end up putting pressure on eachother. Conduction occurs slower in gases than it would in a solid, because the particles in gas runs into eachother less often. There would be way more conduction in metals than in other objects because it has electrons that carry thermal energy. The electrons would obviously run into eachother.
-Conduction energy is a lot like what happens within a material or between two things that are touching. Mostly in solids, conduction occurs when materials vibrate. In Newtons Cradle you pull one ball up and release it to hit the rest of the balls. When you do this you might expect all the balls to move back and forth, but all that happens is one ball is repelled on the other side. What happpens is the kinetic energy (energy of motion) moves from one ball to the last one. Conduction is similar because collision in particles transfer thermal energy without transfering matter.
-Conduction happens when two things are together and they end up putting pressure on eachother. Conduction occurs slower in gases than it would in a solid, because the particles in gas runs into eachother less often. There would be way more conduction in metals than in other objects because it has electrons that carry thermal energy. The electrons would obviously run into eachother.
-Conduction energy is a lot like what happens within a material or between two things that are touching. Mostly in solids, conduction occurs when materials vibrate. In Newtons Cradle you pull one ball up and release it to hit the rest of the balls. When you do this you might expect all the balls to move back and forth, but all that happens is one ball is repelled on the other side. What happpens is the kinetic energy (energy of motion) moves from one ball to the last one. Conduction is similar because collision in particles transfer thermal energy without transfering matter.
Convection
-Convection- transfer of thermal energy when particles of a fluid move from one place to another
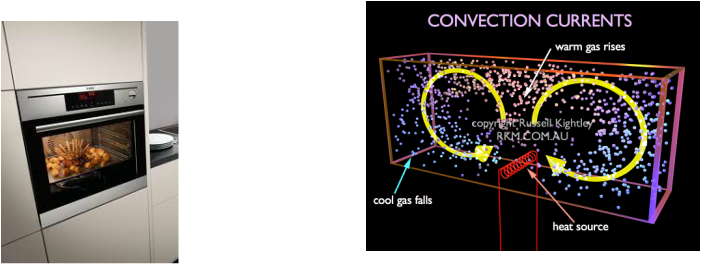
-Example of convection would be hot air rising up when your cupcakes are baking in the oven.
- Have you ever wondered why the temperature at the bottom of an oven is higher than the temperature at the top? This is because when the bottom of the oven heats up it expands and becomes less dense than the surrounding air. The heat rises and in the process it becomes cooler which makes the top of the oven colder. This is why you should always bake your brownies at the top of the oven!:D
-Example of convection would be hot air rising up when your cupcakes are baking in the oven.
- Have you ever wondered why the temperature at the bottom of an oven is higher than the temperature at the top? This is because when the bottom of the oven heats up it expands and becomes less dense than the surrounding air. The heat rises and in the process it becomes cooler which makes the top of the oven colder. This is why you should always bake your brownies at the top of the oven!:D
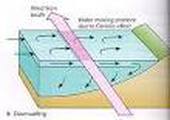
Examples of Convection Currents
-convection currents- occurs when a fluid circulates in a loop as it alternately heats up and cools down.
Radiation
-Radiation- The transfer of energy by waves moving through space
-This type of energy is the reason why we feel hot when the sun is shining on us. The sun can not give us thermal energy (heat) by conduction or convection because there is no air in between the sun and Earth where the heat can be transfered. We get the heat from the sun by waves. This is how you would get your tan on the beach during the summer.
-This type of energy is the reason why we feel hot when the sun is shining on us. The sun can not give us thermal energy (heat) by conduction or convection because there is no air in between the sun and Earth where the heat can be transfered. We get the heat from the sun by waves. This is how you would get your tan on the beach during the summer.
- The sun is actually not the only thing that radiates energy. All objects radiate energy, and the farther away you go from a radiated object the less radiation you will get from it.
Thermodynamics
-Thermodynamics- the study of the conversions between thermal energy and other forms of energy.
Laws of Thermodynamics
1.) Energy is conserved.
2.) Thermal energy can flow from colder objects to hotter ones only if work is done in a system.
3.) Absolute zero can not be reached.
-As you already know energy can not be created or destroyed, but it can be converted into different forms. For example, when starting a fire you are using energy to try to get it going. When the fire gets going it creates thermal energy from the heat and then turns into radiation when the fire warms you up.
Laws of Thermodynamics
1.) Energy is conserved.
2.) Thermal energy can flow from colder objects to hotter ones only if work is done in a system.
3.) Absolute zero can not be reached.
-As you already know energy can not be created or destroyed, but it can be converted into different forms. For example, when starting a fire you are using energy to try to get it going. When the fire gets going it creates thermal energy from the heat and then turns into radiation when the fire warms you up.
-Thermal energy flows spontaniously from hot objects to cold ones. We know this because if you eat a cherry popsicle outside during the summer it will melt because of the heat outside. According to the second law of thermodynamics: Thermal enery can flow from colder objects to hotter ones only if work is done in a system. Ways to get a hotter object cool is using something like a freezer which requires work so it can freeze things.
-Waste heat- Thermal energy that is not converted into work (This work is lost to the surrounding enviorment)
-We could make a heat engine work perfectly if the colder weather on the outside of it was at absolute zero. This is never able to happen according to the third law of thermodynamics, because it is impossible to get the temperature to absolute zero. Scientists have almost got matter cooled to that temperature, but they are not quite at that there yet.
-We could make a heat engine work perfectly if the colder weather on the outside of it was at absolute zero. This is never able to happen according to the third law of thermodynamics, because it is impossible to get the temperature to absolute zero. Scientists have almost got matter cooled to that temperature, but they are not quite at that there yet.